Recent News
- Effective combinatorial drug therapy for prostate cancer September 13, 2022
Prostate cancer is the second most frequent solid organ malignancy in males worldwide. The risk of causing prostate cancer is increased by age, race, and family history. The U.S. FDA has approved the six most successful drugs, viz., docetaxel, sipuleucel-T, abiraterone, enzalutamide, cabazitaxel, and radium-223. Despite these approved therapies, the disease state remains lethal. The recent publication of Dr Imran Uddin, Post Doctoral Fellow, Department of Physics, “Targeted non-AR mediated smart delivery of abiraterone to the prostate cancer” proposes a combinatorial system against prostate cancer using the FDA-approved drug abiraterone. The paper was published in the Q1journal PLoS ONE having an Impact Factor of 3.75. The research was done in collaboration with Dr Mohd Sajid Khan, Associate Professor, Aligarh Muslim University.
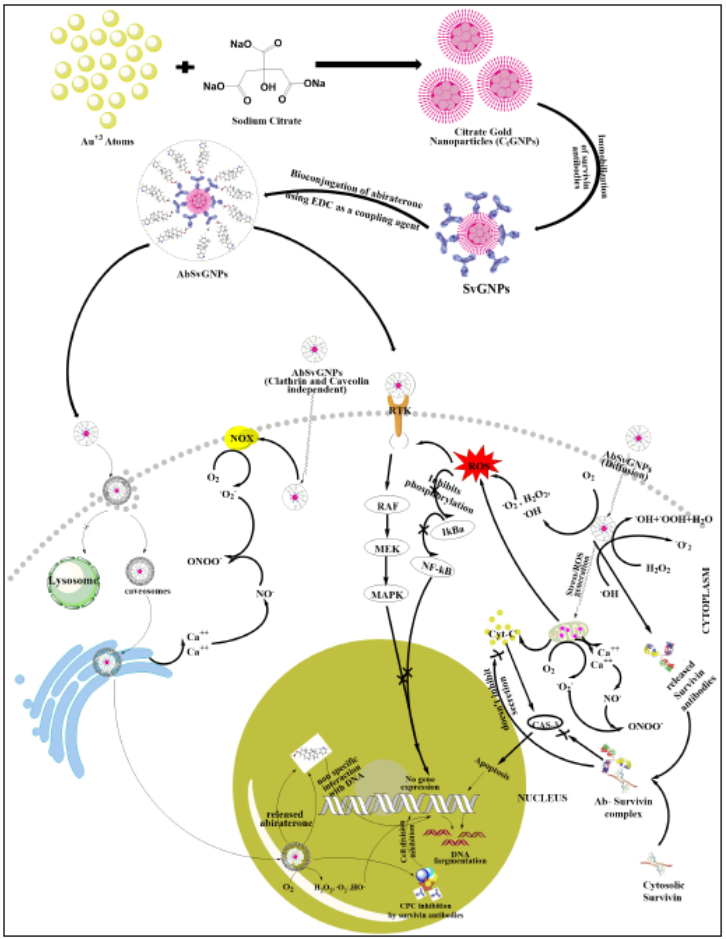
Although abiraterone is an excellent anticancer agent, it causes several side effects and becomes irresponsive after a few months of therapy. They developed a nanomedicine, along with two other components, that will deliver a substantially small dose of abiraterone for treating the same stage of cancer, and the drug will also not be resistant to the cancer cells. The delivery system delivered the drug at a specific site and modified its mode of action. The low dose of abiraterone will also not cause any substantial side effects. The combo was found to be highly biocompatible, nontoxic, and effective.
The proposed nanomedicine with established drug abiraterone, gold nanoparticles, and antibodies against cancer-promoting protein synergistically acted on prostate cancer cells. This synergism potentiated the effect of abiraterone at a very low concentration because other entities also acted via different routes and weakened the cancer cells. The low dose minimized the side effects and maintained patient compliance. This drug was delivered directly to the target, which enabled it to adopt different methods to act on cancer cells. Therefore, the results were promising but further needed to be validated in pre-clinical and clinical studies.
In future, Dr Uddin intends to focus on interdisciplinary sciences. His plans include studying the interface of biology with inorganic nanomaterials, understanding the underlying biological process, and developing new industrially relevant nanomaterials and biomedical aspects. It involves developing nano biosensors for biomolecule detection through the effective integration of the best approaches and expertise in sensor engineering with the vision to take a lead in shaping the future of biomedical monitoring systems. The timely integration of such interdisciplinary approaches will consolidate the application of Lab-on-a-Chip devices for automated biomolecular monitoring.
Abstract of the Research
Prostate cancer is the second-deadliest tumour in men all over the world. Different types of drugs with various delivery systems and pathways were developed, but no one showed prominent results against cancer. Meanwhile, nanotechnology has shown good results against cancer. Therefore, in the given study, citrate mediated synthesized gold nanoparticles (CtGNPs) with immobilized survivin antibodies (SvGNPs) were bioconjugated to the substantially potent drug abiraterone (AbSvGNPs) to develop as a combinatorial therapeutic against prostate cancer. The selected drug abiraterone possesses exceptionally good activity against prostate cancer, but cancer cells develop resistance against this drug and it also poses several severe side effects. Meanwhile, survivin antibodies were used to deliver AbSvGNPs specifically into cancer cells by considering survivin, an anti-apoptotic overexpressed protein in cancer cells, as a marker. The surviving antibodies have also been used to inhibit cancer cells as an immunotherapeutic agent. Similarly, CtGNPs were discovered to inhibit cancer cell proliferation via several transduction pathways. The given bioconjugated nanoparticles (AbSvGNPs) were found to be substantially effective against prostate cancer cells.
Continue reading → - Studying the critical behaviour in physical systems through inequality analysis September 9, 2022
Dr Soumyajyoti Biswas, Assistant Professor from the Department of Physics, has been keenly involved in intense research around areas like the statistical physics of fracture and breakdown in disordered materials and machine learning methods in predicting the imminent breakdown in disordered systems. He has recently published two articles titled “Success of social inequality measures in predicting critical or failure points in some models of physical systems” and “Evolutionary Dynamics of Social Inequality and Coincidence of Gini and Kolkata indices under Unrestricted Competition” in the journals Frontiers in Physics and International Journal of Modern Physics C respectively. The research was done in collaboration with various academicians and undergraduate students (BTech CSE and BSc Physics) from the Indian Statistical Institute, Kolkata and Saha Institute of Nuclear Physics, Kolkata.
It is known that physical systems behave erratically near critical points. Since the 1970s, the ‘erratic’ behaviour has been explained in terms of critical phenomena, and it was found that there are some robust patterns in classes of systems, e.g., all liquid-gas transitions have something in common. Those common patterns were quantified in terms of critical exponents – some numbers that belong to a particular class of systems.
The research shows that if the ‘erratic’ responses of systems near critical points are quantified by some measures of inequality indices (higher the values of the indices, higher the inequality), then such indices behave in a near-universal way for different physical systems, even if they belong in different universality classes. The articles have shown such behaviour in models of physical systems. They have also shown that in socio-economic data, which are also the systems that were conjectured to be in the self-organized critical state. The behaviour from real data matches very well with those from the model simulations.
The researchers have tested their observations from the model simulations to various socio-economic systems that were long conjectured to be in the state of self-organized criticality. Specifically, they have looked into the income inequalities in the US, inequality in citations of authors, inequality in income from movies, and inequality in fluctuations of Bitcoin markets. In all these systems, the participating agents compete among themselves without much external intervention.
In fact, the only system among these where there are some interventions is income inequality. They have shown that through data from the IRS in the US, that inequality has consistently grown in the 1980s till date and has been following the path predicted in our model simulations.
In future, they plan on continuing along this line of looking at critical behaviour in physical systems through inequality analysis. Particularly for the systems where the critical point can represent a catastrophic event (say, fracture) and it is important to quantify the distance from such a catastrophic point.
Abstract of the Research
In many physical systems, experimentally measurable quantities vary drastically near the critical point of such systems. For example, in liquids turning into gas, the densities fluctuate, similar fluctuations happen for magnetisation near critical temperature. We have shown that in systems where the critical point is self-organized i.e., the system reaches the critical point on its own, the unequal nature of their responses show nearly universal trends, even if the models belong to different universality class. This observation could then be used in physical and also socio-economic systems, to quantify their distance from critical point.
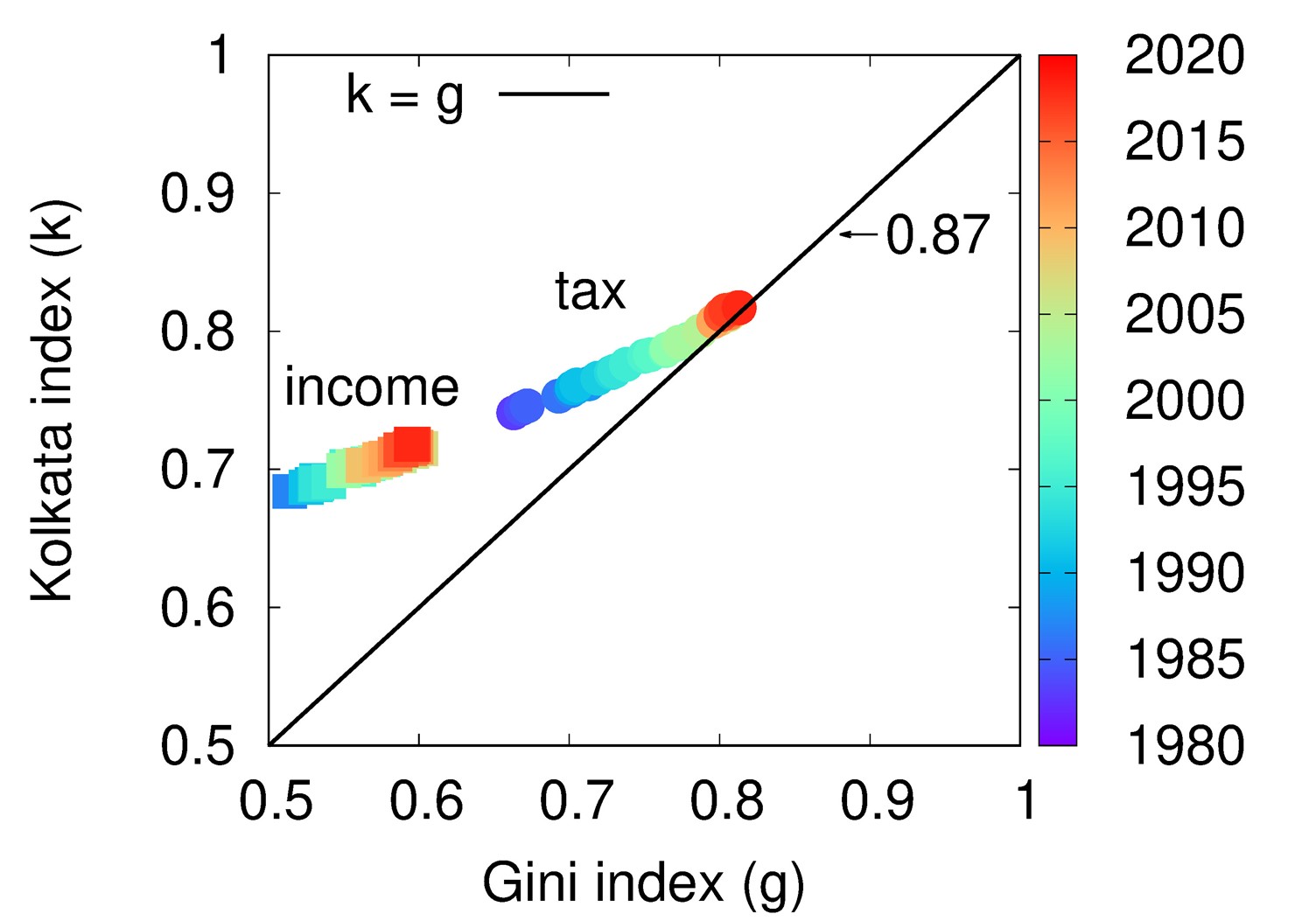
Continue reading →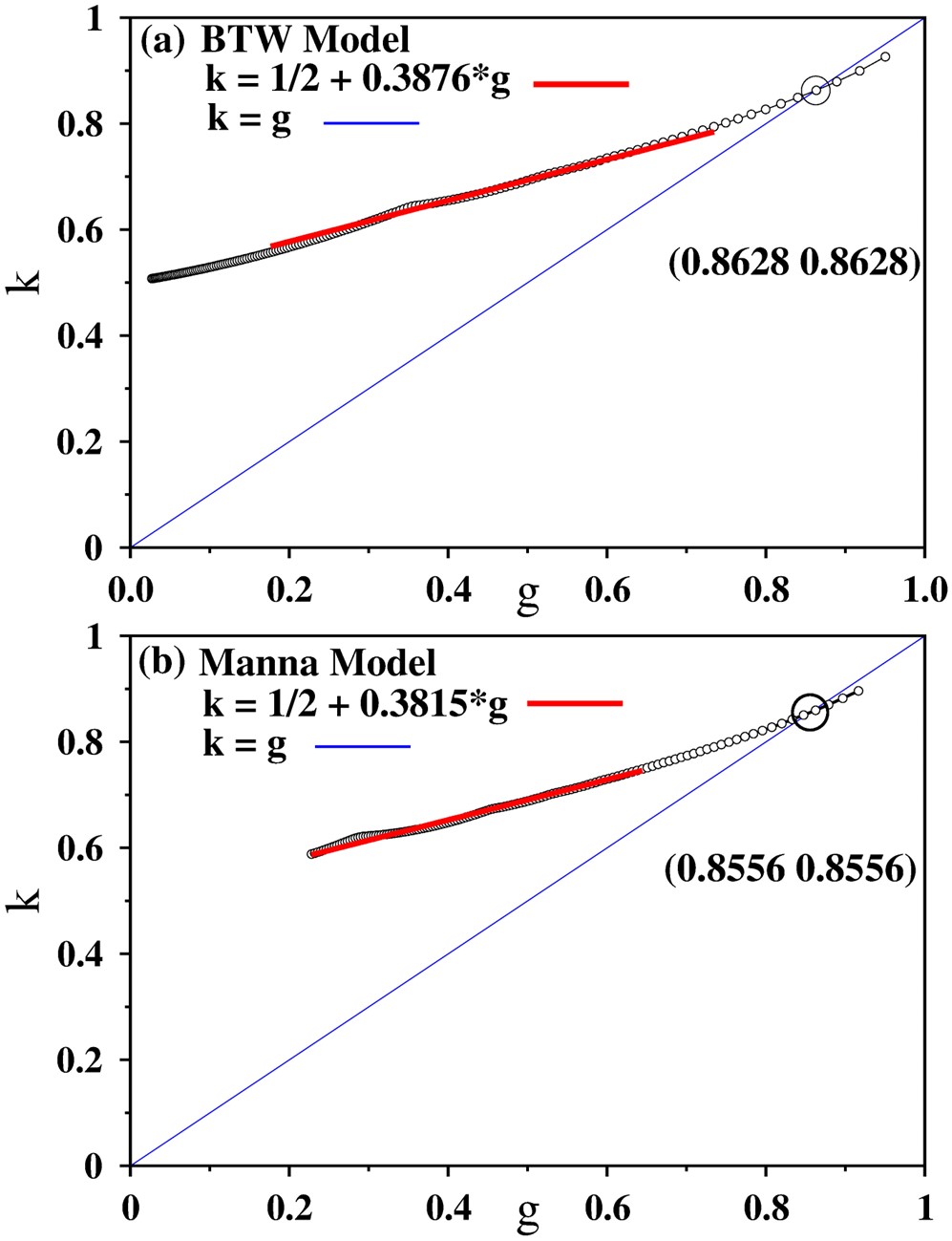 The right hand side figure illustrates the variation of the inequality indices and the circle indicates the critical point where the system is evolving towards. On the left hand side, the picture presents the same indices for income inequality in the US. It has been observed that the inequality has grown over the years and tending towards the saturation value (about 0.86) in a very similar way that is seen in models.
The right hand side figure illustrates the variation of the inequality indices and the circle indicates the critical point where the system is evolving towards. On the left hand side, the picture presents the same indices for income inequality in the US. It has been observed that the inequality has grown over the years and tending towards the saturation value (about 0.86) in a very similar way that is seen in models. - Large-scale production of BP nanosheets September 1, 2022
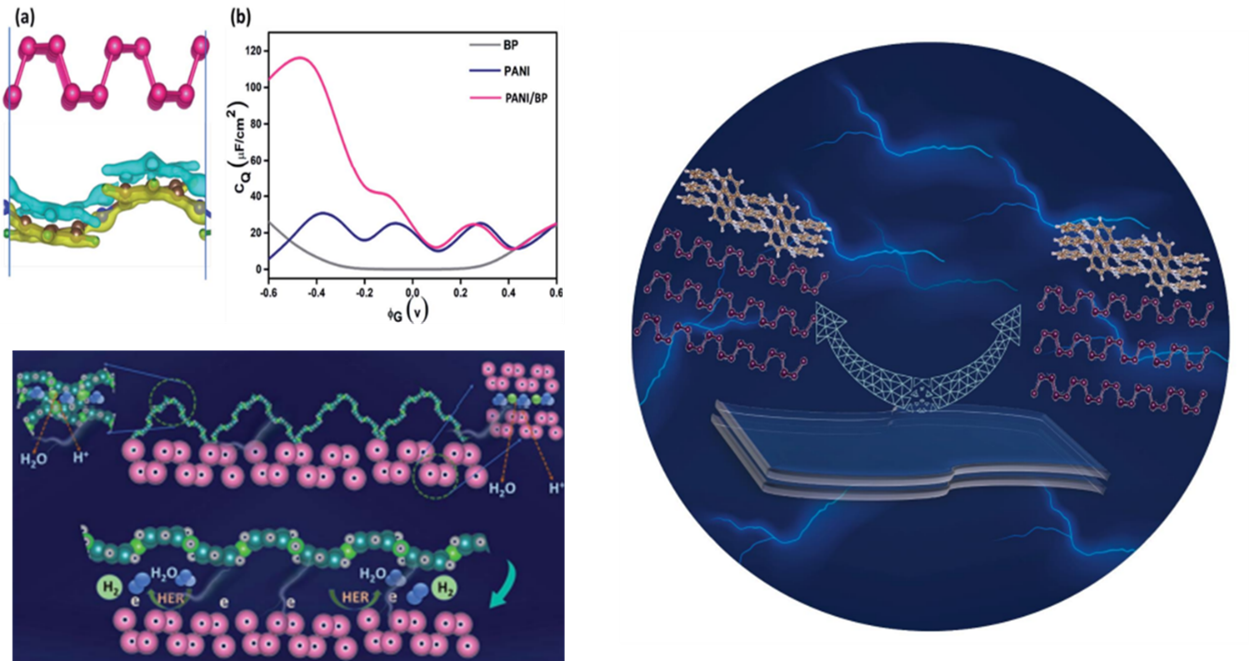 Research at the Department of Physics has effectively produced and characterised BP nanosheets on a large scale by a simple solvothermal approach, and the formation mechanisms are discussed. The paper, 2D-Black Phosphorus/Polyaniline Hybrids for Efficient Supercapacitor and Hydrogen Evolution Reaction Applications Check for updates, has been published by Prof Ranjit Thapa, Associate Dean of Sciences, as a corresponding author, and his PhD student, Mr Samadhan Kapse in Sustainable Energy & Fuels having an Impact Factor of 6.367.
Research at the Department of Physics has effectively produced and characterised BP nanosheets on a large scale by a simple solvothermal approach, and the formation mechanisms are discussed. The paper, 2D-Black Phosphorus/Polyaniline Hybrids for Efficient Supercapacitor and Hydrogen Evolution Reaction Applications Check for updates, has been published by Prof Ranjit Thapa, Associate Dean of Sciences, as a corresponding author, and his PhD student, Mr Samadhan Kapse in Sustainable Energy & Fuels having an Impact Factor of 6.367.Abstract
Black phosphorous (BP) is an emerging 2D material with exciting physicochemical properties with broad applicability in electronics. Stability in the ambient environment, large-scale synthesis, and volume expansion during the charge/discharge process hinder its application in energy storage. Here, we report a facile gram-scale synthesis of BP in a mild reaction condition by a simple and cost-effective wet chemical method. To overcome its degradation and sluggish electrochemical performance, an organic hybrid with polyaniline is also prepared. Further, we fabricated a flexible supercapacitor device which results in an exceptional specific capacitance of 969 mFcm-2 at a current density of 0.4 Acm-2, which displayed a high energy density of 21.5 mWhkg-1 at a power density of 231 mWkg-1 with good cycling stability of 91% after 4000 charge-discharge cycles. Similarly, the cyclic voltammetry studies of the flexible devices at various bending angles display a similar CV profile for all the bending angles, which confirms the device’s reliability for flexible applications.
Explanation of the research
BP-PANI hybrid materials were prepared by the in-situ chemical oxidation method. By this approach, the researchers got highly stable BP by an inorganic-organic linkage, and its energy storage performance was also investigated. The fabricated symmetric flexible supercapacitor device based on BP/PANI heterostructure exhibited an extraordinary specific capacitance of 969 mFcm-2 at a current density of 0.4 Acm-2. Moreover, the fabricated device showed a high energy density of 21.5 mWhkg-1 and a power density of 231 mWkg-1 with impressive cycle stability of 91% after 4000 charge-discharge cycles. This study paves the way for future research into gram-scale BP synthesis, stability via an inorganic-organic coupling, and its potential application in electrochemical energy storage devices.
Social implications of the research
With the rapid growth of portable/flexible electronics and the high demand for clean energy, supercapacitors have sparked interest due to their advantages of fast charge/discharge rates, long cycle life, and high-power density compared to conventional energy-storage devices such as dielectric capacitors and Li-ion batteries. Likewise, developing new functional materials with outstanding properties could shed light on many issues, including pollution, energy, synthesis, and cost. In recent years few graphene analogues materials have been explored, and because of their tuneable physicochemical properties, they were used in energy storage applications. Generally, black phosphorus was synthesised from polymorphs of phosphorus under vigorous reaction conditions. However, these high temperature/pressure conditions suffer from safety, toxicity, controllability, and gram-scale production.
Quantum capacitance is an efficient tool for rapidly screening materials for supercapacitor applications and therefore is the future of this research. The researchers have collaborated with Mr Namsheer K, Mr Mridula Manoj, Mr Aditya Sharma, and Dr Chandra Sekhar Rout from the Functional Materials & Devices Laboratory, Centre for Nano Material Sciences, Jain University, Bangalore, India, in this work.
Continue reading → - Efficient and selective single-atom catalysts for eNRR September 1, 2022
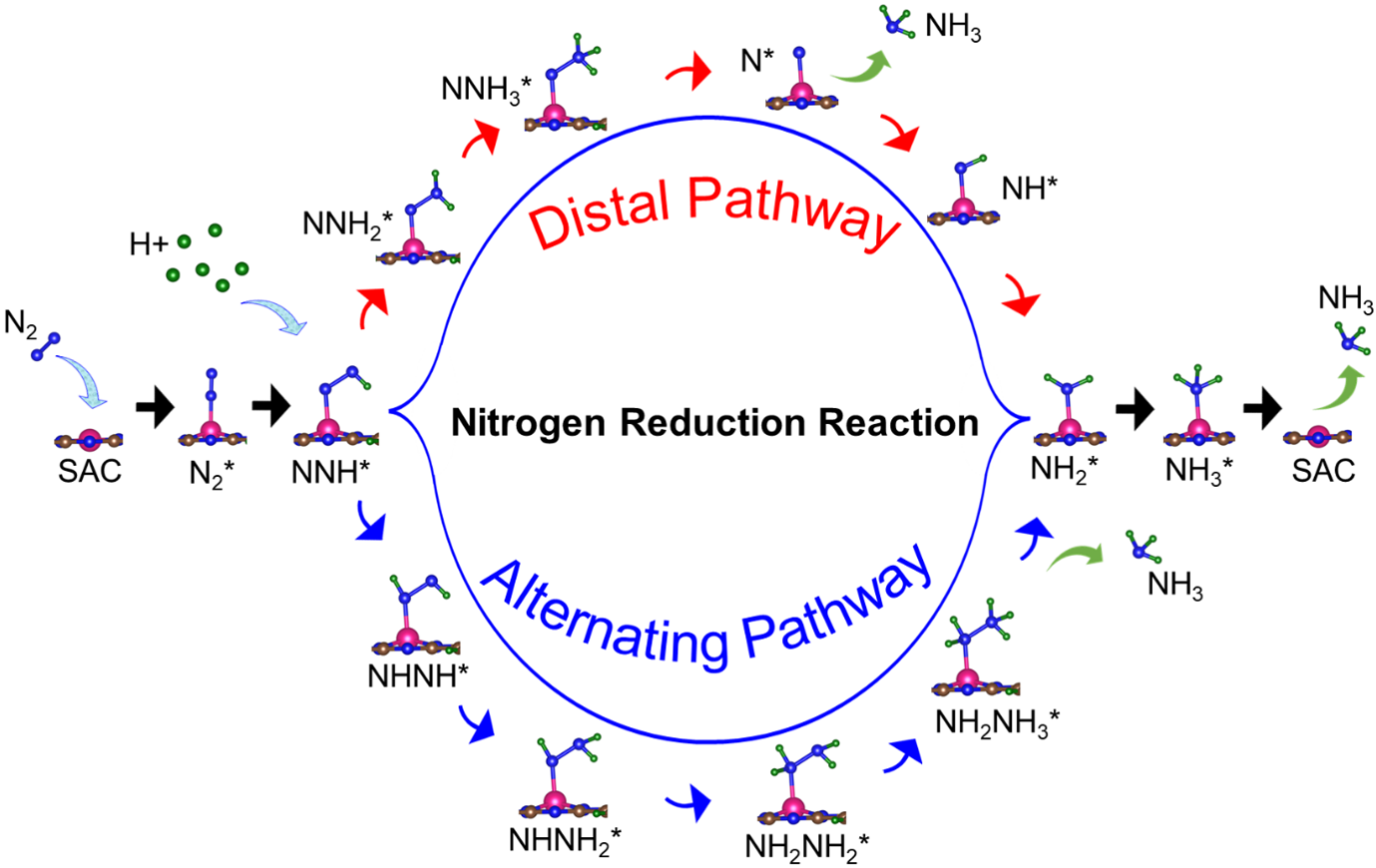
The Department of Physics is glad to announce that Dr Ranjit Thapa and his PhD scholar Mr Samadhan Kapse have published their research paper “Descriptors and graphical construction for in silico design of efficient and selective single-atom catalysts for eNRR” in the journal Chemical Science, having an Impact Factor of 9.969. The paper was published in collaboration with Prof Shobhana Narasimhan, Theoretical Sciences Unit and School of Advanced Materials, Jawaharlal Nehru Centre for Advanced Scientific Research, Bangalore. Chemical Science is a highly prestigious nature Index journal, which accepts only breakthrough research contributions for publication.
The Haber-Bosch process for ammonia synthesis has been described as possibly the most important scientific discovery of the twentieth century. However, it requires high temperatures and pressures and results in large energy consumption and emission of greenhouse gases. That is where electrochemical nitrogen reduction reaction (eNRR) comes into the picture. It synthesizes ammonia from nitrogen and water under mild conditions (N2 + 6H+ + 6e- → 2NH3). However, currently available eNRR catalysts need improvement in three respects: (i) the efficiency of nitrogen fixation needs to be increased, (ii) the competing hydrogen evolution reaction (HER) needs to be suppressed, and (iii) hydrogen poisoning of active sites must be avoided. Transition metals are popular eNRR catalysts; however, they tend to favour hydrogen adsorption due to the formation of strong metal d – hydrogen σ bonds, and tend to have a low affinity for N2 adsorption. Their research mitigates these problems by appropriately tuning the electronic structure by altering the environment surrounding metal atoms at the active site of single-atom catalysts (SACs). Moreover, in previous works, typically, only one criterion (usually competing HER) was used to optimize catalyst function, whereas they simultaneously optimised the catalyst function with respect to multiple criteria.
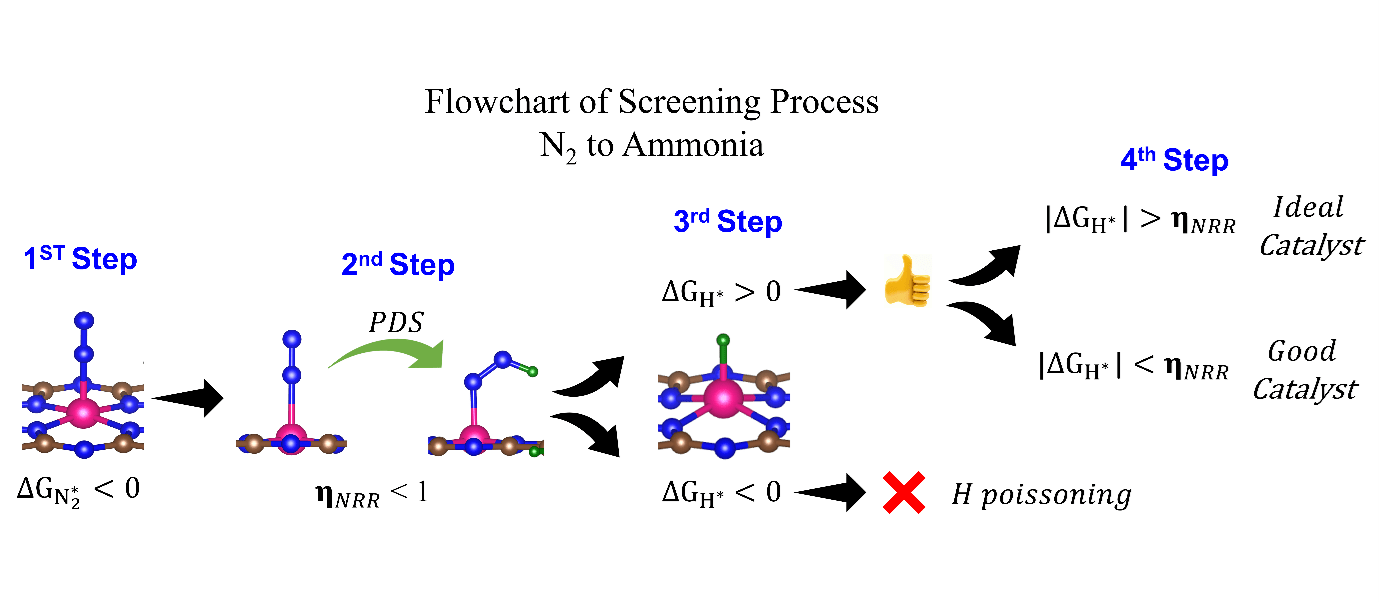
They have screened 66 different transition metal-based SACs for possible use in eNRR. To determine the best possible catalyst, they considered three factors: N2 adsorption, hydrogen poisoning and the overpotential of eNRR. Here, the valence electron occupancy (Oval) is identified as a new electronic descriptor that can predict the overpotential value. They emphasised that having a low η_NRR alone does not suffice to indicate a suitable eNRR catalyst, since if the adsorption free energy is higher for H than N2, active sites will be poisoned, hindering eNRR. Thus, they present a simple graphical procedure for identifying the most promising catalysts. To carry out this procedure, one must compute only 〖ΔG〗_(H^* ) and 〖ΔG〗_(NNH^* ), the changes in the free energies of H and NNH adsorption, respectively (note that η_NRR can be deduced if 〖ΔG〗_(NNH^* ) is known). The most promising candidate is identified as Sc-Pc, which they predict will have no H poisoning and will be highly selective for eNRR over HER. Moreover, they predict that Mn-Pc, Cr-N4, Fe-N2C2 should also be highly efficient, with low overpotential (η_NRR < 1 V) toward eNRR, and no H poisoning. In future they aim to find the selective materials for catalytic reactions by studying the origin of activity, reaction mechanism, etc.
Abstract of the Research
The electrochemical nitrogen reduction reaction (eNRR) offers the possibility of ammonia synthesis under mild conditions; however, it suffers from low yields, a competing hydrogen evolution reaction pathway, and hydrogen poisoning. We present a systematic approach toward screening single atom catalysts (SACs) for eNRR, by focusing on key parameters computed from density functional theory, and relationships between them. We illustrate this by application to 66 model catalysts of the types, TM-Pc, TM-NXCY, and TM-N3, where TM is a 3d transition metal or molybdenum. We identified the best SACs as Sc-Pc, Cr-N4, Mn-Pc, and Fe-N2C2; these show eNRR selectivity over HER and no hydrogen poisoning. The catalysts are identified through multi-parameter optimization which includes the condition of hydrogen poisoning. We propose a new electronic descriptor Oval, the valence electron occupancy of the metal center, that exhibits a volcano-type relationship with eNRR overpotential. Our multi-parameter optimization approach can be mapped onto a simple graphical construction to find the best catalyst for eNRR over HER and hydrogen poisoning.
- VS2-BP hybrid electrode material for supercapacitor applications August 24, 2022
 A theoretical investigation is highly important to investigate the properties of materials, the origin of selectivity, and the effect of various parameters in designing promising electrode materials for supercapacitor applications. The latest research paper by Mr Samadhan Kapse, PhD Student in the Department of Physics, and Prof Ranjit Thapa, Associate Dean of SEAMS (Sciences), envisions this and developed a novel VS2-BP hybrid electrode material. Their article titled All-solid-state Supercapacitor Based on Advanced 2D Vanadium disulfide/Black Phosphorus Hybrids for Wearable Electronics has been published in the journal ACS Applied Energy Materials with an impact factor of 6.959.
A theoretical investigation is highly important to investigate the properties of materials, the origin of selectivity, and the effect of various parameters in designing promising electrode materials for supercapacitor applications. The latest research paper by Mr Samadhan Kapse, PhD Student in the Department of Physics, and Prof Ranjit Thapa, Associate Dean of SEAMS (Sciences), envisions this and developed a novel VS2-BP hybrid electrode material. Their article titled All-solid-state Supercapacitor Based on Advanced 2D Vanadium disulfide/Black Phosphorus Hybrids for Wearable Electronics has been published in the journal ACS Applied Energy Materials with an impact factor of 6.959.Abstract
Vanadium disulfide-Black Phosphorus (VS2-BP) hybrids were synthesised by a one-pot hydrothermal assisted method to achieve enhanced electrochemical activity for supercapacitor applications. The concentration of BP was optimised to prevent the restacking nature of VS2 and to enrich the active edges for electrolytic ion intercalation. The charge storage kinetics of the best-performing VS2-BP as an active electrode has demonstrated the dominance of the pseudocapacitive nature of the material. Further, by sandwiching with PVA/K2SO4 gel electrolyte, an all-solid-state (ASS) Vanadium disulfide/Black Phosphorus-50 mg (VS2-BP-50) symmetric device was developed on highly conductive carbon paper. The ASS VS2-BP-50 symmetric device displays the highest specific areal capacitance of 203.25 mF/cm2. It exhibits the maximum areal energy density of 28.22 µW h cm-2 at an areal power density of 596.09 mW cm-2, outperforming previous literature. We used density functional theory to understand the origin of high quantum capacitance. We found that the charge accumulation region between VS2 and BP monolayers and the charge transfer is the origin of the improved density of states in the VS2-BP hybrid. Also, we observed the higher mobility of K+ ion and a higher diffusion rate using the Density functional theory (DFT) method.
Explanation of the research
A novel VS2-BP hybrid electrode material was prepared using a simple hydrothermal approach. Due to a synergistic effect, it was discovered that adding BP to metallic VS2 enhances the number of electrochemically active sites, resulting in increased surface activity. It also accelerates reaction kinetics with electrolyte ions by improving the electrical behaviour of active electrode material. As a result, the hybrid technique overcomes the weaknesses of individual components during electrochemical processes, resulting in increased performance that has been limited by individuals. The BP nanosheets behaved as a pore region for electron transport and prevented the VS2 layers from re-stacking. Systematic experiments are conducted by selecting the ideal precursor ratios to generate a high-quality VS2-BP hybrid with enhanced electronic conductivity. Furthermore, in the overall collective charge storage of the VS2-BP-50 hybrid material, the present results demonstrated that capacitive contributions outnumber diffusive contributions. The ASS VS2-BP-50 symmetric supercapacitor device was also designed to have a high areal capacitance of 203.25 mF/cm2 with a maximum areal power density of 596.09 mW cm-2. The extraordinary performance of the ASS VS2-BP-50 symmetric device illustrates its versatility in terms of designing a high-power density ASS supercapacitor for flexible and wearable device applications. The work functions of BP, VS2, and VS2-BP are 0.73 eV, 5.37 eV, and 4.99 eV, respectively, which help in the charge transfer mechanism and increase the density of state at the Fermi level, and subsequently, the quantum capacitance of the heterostructure.
Collaborations
1. Mr Aditya Sharma, Centre for Nano and Material Sciences, Jain Global Campus, Jakkasandra, Ramanagaram, Bangalore – 562112, Karnataka, India
2. Mr Ankur, Centre for Nano and Material Sciences, Jain Global Campus, Jakkasandra, Ramanagaram, Bangalore – 562112, Karnataka, India
3. Mr Sagar Bisoyi, Department of Physics, School of Applied Sciences, KIIT Deemed to be University, Bhubaneswar-751024, Odisha, India.
4. Dr Gopal K. Pradhan, Department of Physics, School of Applied Sciences, KIIT Deemed to be University, Bhubaneswar-751024, Odisha, India.
5. Dr Chandra Sekhar Rout, Centre for Nano and Material Sciences, Jain Global Campus, Jakkasandra, Ramanagaram, Bangalore – 562112, Karnataka, India
Social implications of the research
With the exponential development of portable/flexible electronics and the high demand for renewable energy, conventional energy-storage devices, such as supercapacitors, have attracted attention due to their benefits of fast charge/discharge rates, long cycle life, and high-power density. Similarly, developing novel functional materials with exceptional qualities could shed light on a plethora of challenges, including environmental pollution, energy crisis, etc. Two-dimensional (2D) layered materials, such as metallic 1T MoS2 single layers, SnSe2, MXenes, and black phosphorous (BP), have been intensively studied for supercapacitor applications. These materials benefit from efficient ion intercalation and electrosorption. The two-dimensional (2D) layered transition-metal dichalcogenides (TMDs) have recently piqued the scientific community’s curiosity.

