Recent News
- Faculty Collaborate on Interdisciplinary Innovation December 3, 2024
 In an inspiring collaboration, Prof. G S Vinod Kumar and Prof. Sheela Singh from the Department of Mechanical Engineering, Prof. Ranjit Thapa from the Department of Physics, and Dr Rajapandiyan Panneerselvam from the Department of Chemistry at SRM University – AP, along with PhD scholars Ms Harsha K and Ms Arunima J, have co-authored a compelling paper titled “Innovative Approaches to Enhancing the Tarnish Resistance of Silver Alloys.” This groundbreaking research focuses on developing new silver alloys that resist tarnishing, enhancing durability and aesthetic appeal for applications in the jewellery industry. Together, they are paving the way for innovative solutions that blend science with artistry.
In an inspiring collaboration, Prof. G S Vinod Kumar and Prof. Sheela Singh from the Department of Mechanical Engineering, Prof. Ranjit Thapa from the Department of Physics, and Dr Rajapandiyan Panneerselvam from the Department of Chemistry at SRM University – AP, along with PhD scholars Ms Harsha K and Ms Arunima J, have co-authored a compelling paper titled “Innovative Approaches to Enhancing the Tarnish Resistance of Silver Alloys.” This groundbreaking research focuses on developing new silver alloys that resist tarnishing, enhancing durability and aesthetic appeal for applications in the jewellery industry. Together, they are paving the way for innovative solutions that blend science with artistry.Abstract
Silver and its alloys undergo tarnishing with time, which is a black stain on the surface due to the formation of Ag2S. Developing a tarnish resistant Ag alloy was attempted by alloying Ag with elements that form a passive oxide layer on the surface. Germanium is proven to provide better tarnish resistance to sterling silver alloy (92.5wt.% pure) which is available under the trade name of Argentium©. The present work investigates the tarnish resistance behaviour of sterling silver alloy (92.5wt.% pure) containing various additions of Copper, Zinc, Germanium, Aluminium, Beryllium, Titanium, Zirconium, and Magnesium. The alloys were prepared by melting and casting route, followed by Passivation Heat Treatment (PHT) to create a stable and continuous oxide layer. The temperature for PHT was optimized using thermogravimetry analysis (TGA) of the alloys prepared. An accelerated tarnish test was carried out to investigate the tarnishing behaviour of alloy samples obtained before and after PHT. The samples were characterized using XRD, SEM-EDX, TG-DSC, micro-Raman Spectroscopy, and XPS. The change in reflectance of the samples after the tarnish test is determined using UV-visible reflectance spectroscopy. The mechanism behind the tarnish resistance was derived using Density Functional Theory (DFT) by comparing sulphur (S2) and Oxygen (O2) adsorption energies (BE) of the alloying elements.
Explanation of the Research in layperson’s terms
Age-old silver pieces are found in different colours ranging from light yellow(silver Jewellery pieces after a few weeks of usage) to black(archaeological silver pieces). They look different in terms of the metallic white colour and lustrous appearance from fresh silver. This demeans silver and thus affects its market. The major cause of the staining of silver is the interaction of individual silver particles with some elements found common in the atmosphere. We work on the prevention of this staining of silver. For the study, silver is incorporated with other elements which makes silver less interacting with staining elements in the atmosphere. These additional elements create a layer over silver so that it is protected. The appropriate elements are identified by using modelling using computer software. After obtaining appropriate elements, the experimental trials are also done with the same elements until stainless silver is obtained. Then, what happens to the added elements inside silver is also studied by using modern microscopic technics.
Practical/Social Implications of the Research
The major application of the invention is in the jewellery industry. The problem of tarnishing is an age-old threat in jewellery making. The alloys we proposed could be used to make quality silver jewellery/articles that can sustain the colour and lustre for a longer period. This will stop the hesitation from jewellery designers and industries to try intricate designs in silver and find a better market for them. The alloys based on the proposed composition show good grain refinement and thus high hardness. This strengthens the soft silver and improves the range of its applications from low hardness articles to high. The alloys based on the proposed composition have high tensile strength. They deform plastically for a wide range of stress values and will not break easily.
Collaborations:
Waman Hari Pethe & Sons Jewellery
Future Research Plans:
1. Corrosion studies of silver alloys to understand the behaviour in solutions having compositions similar to that of sweat.
2. Study of mechanical properties of silver alloys to develop workable alloys of sterling silver which could be used for jewellery manufacturing
3. Identification of elements having better oxide layer formation when alloyed with silver, by using computational techniques and experimental studies of their properties.
Link to the article:
Continue reading →
https://www.sciencedirect.com/science/article/pii/S2238785424024633 - New Theories on Dark Matter: Dr Basabendu Proposes New Insights in Q1 Journal October 4, 2024
 In a significant advancement in the field of astrophysics, Dr Basabendu Barman, Assistant Professor in the Department of Physics, has published a groundbreaking paper titled “Dark matter-electron scattering and freeze-in scenarios in the light of \textit{Z’} mediation” in the prestigious Q1 journal, Physical Review D, known for its impact factor of 5.0 within the Nature Index.
In a significant advancement in the field of astrophysics, Dr Basabendu Barman, Assistant Professor in the Department of Physics, has published a groundbreaking paper titled “Dark matter-electron scattering and freeze-in scenarios in the light of \textit{Z’} mediation” in the prestigious Q1 journal, Physical Review D, known for its impact factor of 5.0 within the Nature Index.Dr Barman’s research delves into the enigmatic realm of dark matter, proposing a novel mechanism through which dark matter might interact with the visible universe via a hypothesised fifth force. This interaction could provide vital insights into the nature of dark matter and its elusive characteristics. The study suggests that if this interaction is confirmed, experiments exploring the existence of a fifth force could concurrently unravel the mysteries surrounding dark matter, leading to a deeper understanding of the universe.
This publication not only highlights the potential for new discoveries in particle physics but also positions SRM University-AP at the forefront of cutting-edge research in dark matter studies. Dr. Barman’s findings could pave the way for further exploration and experimental validation, ultimately contributing to the ongoing quest to decode one of the universe’s greatest mysteries.
Abstract:
We investigate dark matter (DM-)electron scattering in a minimal U〖(1)〗_X extension of the Standard Model (SM), where the DM can appear as a Majorana fermion, a complex singlet scalar, or a Dirac fermion. To study bounds on the new gauge coupling and new gauge boson mass, from DM-electron scattering, we consider several direct search experiments like CDMS, DAMIC, SENSEI, PandaX-II, DarkSide-50, and XENON1T-S2 for different U〖(1)〗_X charges. In this setup, we consider DM production via freeze-in in both radiation-dominated and modified cosmological backgrounds to project sensitivities onto coupling vs mass plane satisfying observed relic abundance. DM-electron scattering could provide comparable, or even stronger, bounds compared to those obtained from the electron/muon (g-2), low-energy scattering, and intensity frontier experiments within mass range of 0.01-0.1 GeV. Constrains from freeze-in could provide stronger sensitivities for new gauge boson masses above about 1 GeV ; however, these limits are comparable to those obtained from LHCb and LEP experiments for mass between 10-150 GeV. In the future, electron-muon scattering (MUonE), proton (FASER and DUNE), and electron/positron (ILC) beam-dump experiments could probe these parameters.
From Layperson’s perspective:
We know there are four fundamental forces in nature: strong, weak, electromagnetic, and gravitational. But what if there’s a hidden, fifth force we haven’t discovered yet? The Standard Model of particle physics, which organizes all known particles, doesn’t include this fifth force (and doesn’t include gravity either, unfortunately). So, how can we theoretically create a particle physics model for this possible fifth force? That’s what we explore in this paper. Interestingly, there are already experiments (for example, the Large Hadron Collider or LHC at CERN, Geneva) searching for signs of fifth forces. If this force exists, our model could be tested by these experiments. But there’s more! We also wanted to tackle a big mystery in cosmology: dark matter. Dark matter makes up about 24% of the universe, but we’ve never directly detected it because it doesn’t reflect light—it’s “dark.” However, there are smart ways to try to find it. In this paper, we propose how the same dark matter might interact with the visible universe through this fifth force and thereby leave their footprints. If true, therefore, the experiments looking for the fifth force could also give us clues about the nature of dark matter.
Title:
Dark matter-electron scattering and freeze-in scenarios in the light of Z’ mediation.
In BibTeX (citation) format:
@article{PhysRevD.110.055029,
title = {Dark matter-electron scattering and freeze-in scenarios in the light of ${Z}^{\ensuremath{‘}}$ mediation},
author = {Barman, Basabendu and Das, Arindam and Mandal, Sanjoy},
journal = {Phys. Rev. D},
volume = {110},
issue = {5},
pages = {055029},
numpages = {20},
year = {2024},
month = {Sep},
publisher = {American Physical Society},
doi = {10.1103/PhysRevD.110.055029},
url = {https://link.aps.org/doi/10.1103/PhysRevD.110.055029}
}Practical Implementations & Social Impact:
The primary goal of this study is to explore what lies beyond the known, visible universe. This curiosity has driven humanity for centuries—to build rockets and explore outer space and to smash particles in colliders, searching for the mysteries hidden within the atom. The desire to uncover the unknown, to shed light on the darkness, is a fundamental part of what it means to be human. In this sense, the work contributes to the realm of pure intellectual pursuit. Science speaks the language of data, and data is born from experiments. The validation of any well-constructed theory ultimately depends on experimental evidence. For this reason, it is essential for society to cultivate a culture that values fundamental scientific discussion and increases funding for basic research.
Collaborations:
This work was done in collaboration with Prof. Arindam Das from the Department of Physics, Hokkaido University, Sapporo, Japan, and Dr. Sanjoy Mandal from the Korea Institute for Advanced Study (KIAS), Seoul, Korea.
Future plans:
- A closer look into early universe dynamics by performing more involved simulations.
- Connection between particle physics models and early Universe cosmology.
- Complementary searches from different experiments in unravelling new physics beyond the Standard Model.
- Searching new physics at energy and intensity frontier.
- Dr Nilakantha Meher’s Research Uses Light to Improve Object Detection Precision September 5, 2024

Dr Nilkantha Meher, an Assistant Professor in the Department of Physics at SRM University-AP, has significantly contributed to science with his research paper on using thermal light to detect objects with unmatched precision. This phenomenal work that featured in the journal Physical Review A will positively contribute to the fields of sensing, gravitational wave detection, and phase microscopy.
Abstract:
Estimation of the phase delay between interferometer arms is the core of transmission phase microscopy. Such phase estimation may exhibit an error below the standard quantum (shot-noise) limit, if the input is an entangled two-mode state, e.g., a N00N state. We show, by contrast, that such supersensitive phase estimation (SSPE) is achievable by incoherent, e.g., thermal, light that is injected into a Mach-Zehnder interferometer via a Kerr-nonlinear two-mode coupler. The phase error is shown to be reduced below, being the mean photon number, by thermal input in such interferometric setups, even for small nonlinear phase-shifts per photon pair or for significant photon loss. Remarkably, the phase accuracy achievable in such setups by thermal input surpasses that of coherent light with the same. Available mode couplers with giant Kerr nonlinearity that stems either from dipole-dipole interactions of Rydberg polaritons in cold atomic gas or from cavity-enhanced dispersive atom-field interactions may exploit such effects to substantially advance the interferometric phase microscopy using incoherent, faint light sources.
Practical Implementation:
The proposed nonlinear interferometer in this research can serve as a robust quantum sensor, making it suitable for a range of applications, including object sensing, gravitational wave detection, and phase microscopy.
Your Collaborations:
Prof. Gershon Kurizki (Weizmann Institute of Science, Israel)
Prof. Tomas Opatrny (Palacky University, Czech Republic)
Dr. Eilon Poem (Weizmann Institute of Science, Israel)
Prof. Ofer Firstenberg (Weizmann Institute of Science, Israel)Future Research Plans:
He is currently investigating the sensing of quantum entanglement and generating highly nonclassical states using various nonlinear interferometers. This research has significant implications for distributed quantum communication and quantum information processing.
Continue reading → - Paper on Deciphering Oxygen Evolution Reaction Activity: A QM/ML Approach with Single Atom Catalysts September 5, 2024

Prof. Ranjith Thapa in collaboration with two of his research scholars, Mr E. S. Erakulan Mr Sourav Ghosh and has come up with a groundbreaking research that has resulted in the publication of a scholarly paper titled, Specific Descriptor for Oxygen Evolution Reaction Activity on Single Atom Catalysts Using QM/ML.
Abstract of the paper
Descriptors are properties or parameters of a material that is used to explain any catalytic activity both computationally and experimentally. Such descriptors aid in designing the material’s property to obtain efficient catalyst. For transition metals, d-band center is a well-known descriptor that shows Sabatier type relation for several catalytic reactions. However, it fails to explain the activity when considering same metal active site with varying local environment. To address this, density functional theory was used for single atom catalysts (SACs) embedded on armchair and zigzag graphene nanoribbons (AGNR and ZGNR). By varying the anchoring nitrogen atoms’ orientation and considering pristine and doped cases, 432 active sites were used to test the oxygen evolution reaction (OER) activity. It was observed that S and SO2 dopant helps in reducing the overpotential on Co-SAC (h = 0.28 V). Along with the d-band center, a total of 105 possible descriptors were individually tested and failed to correlate with OER activity. Further, PCA was employed to narrow down unique descriptors and machine learning algorithms (MLR, RR, SVR, RFR, BRR, LASSO, KNR and XGR) were trained on the two obtained descriptors. Among the models, SVR and RFR model showed highest performance with R2 = 0.89 and 0.88 on test data. This work shows the necessity of a multi-descriptor approach to explain OER catalytic activity on SAC and the approach would help in identifying similar descriptors for other catalytic reactions as well.
Social Implications:
Computational studies have proven to be a vital tool to predict new materials and also assess the behaviour towards various catalytic reactions. They also identify the innate properties of the material which drives the catalytic activity. It helps in designing the material with required property to improve the catalytic activity. Descriptors are such computationally obtained properties/parameters of a material that has a meaningful relation with any catalytic property of a chemical reaction. d-band center, given by Hammer and Norskov in 1995, explained the binding strength of oxygen atom on pure transition metals. The d-band center shows Sabatier type relation with chemical activity and has been widely used to explain the catalytic activity of several reactions since its formulation. The adsorbate state after interaction with delocalized s-states of the metal atom is almost constant while that resulting from d-states interaction, is split into bonding and antibonding states. Hence the s-states were not considered. It is well known that, when the dimensions of a system are lowered the states become narrow and localized. In such systems, the d-band center does not explain the catalytic activity well and it is an open research problem.
Future Projects:
Density functional theory with machine learning approach could further be used and improved on similar SACs from which a predictive model equation could be constructed. Also, the proposed models are open to exploration on other catalytic reactions as well.
The authors thank SRM University-AP and National Super Computing mission for providing the computational facility.
Continue reading → - Patent Granted for Research in Developing Tarnish Resistant Silver Alloys May 7, 2024

Yet another groundbreaking achievement for the researchers at SRM University-AP! Prof. Ranjit Thapa, Dean-Research and Professor, Department of Physics, Prof. G S Vinod Kumar, Professor and Head, Department of Mechanical Engineering and Ms Harsha K, PhD scholar, continue to make their mark in the university’s excellent research legacy by having their patent “Tarnish Resistant Silver Composition and a Process for its Preparation” being granted by the Indian Patent Office. This innovative research team has used density functional theory to explain the tarnishing of silver. Their work also focuses on finding alloying elements that protect silver.
Abstract
The research is on the development of tarnish-resistant silver alloys from an experimental and computational perspective. With time, silver atoms on the surface of the metal undergo sulphidation and form Ag2S molecules. These particles will accumulate to form a layer whose thickness goes beyond 10nm, and then the human eye will start to find a discolouration on the surface of silver, which is tarnish. The stain colour changes from light yellow to dark brown. This reduces the lustre of silver and makes them aesthetically non-pleasing. The silver jewellery and articles manufacturing industry suffers from this tarnishing as it leads to the loss of material and money and ruins intricate designs made of silver. The research study attempts the problem by alloying silver with appropriate elements, which are computationally checked and verified. The team works with alloying elements such as Cu, Zn, Ge, Ti, Zr, Mg, Al, and Be. Along with tarnish resistance, the proposed alloys maintain high reflectance, good hardness, and excellent workability when spinning.

Figure.1 The images of different silver alloys after accelerated tarnish test in as-cast condition (first row) and after undergoing passivation heat treatment at 450°C for 1 hour in the presence of oxygen (second row).
Practical implementation/social implications of the research
- Stainless silver is in demand as customers want their precious metal articles to be kept for a longer time as heirlooms. So, the product that we could develop out of our composition will be making more demand for silver.
- It can increase the market potential of silver.
- It can lead to more innovations in the jewellery industry.
Collaborations
- Waman Hari Pethe
- Ashlyn Chemmannur
- TITAN
The team would continue to work on the scope of research to develop more tarnish-resistant compositions, improve the tensile strength, scratch resistance, surface hardness, and workability of silver alloys and find novel elements which can add to desirable properties of silver.
Continue reading →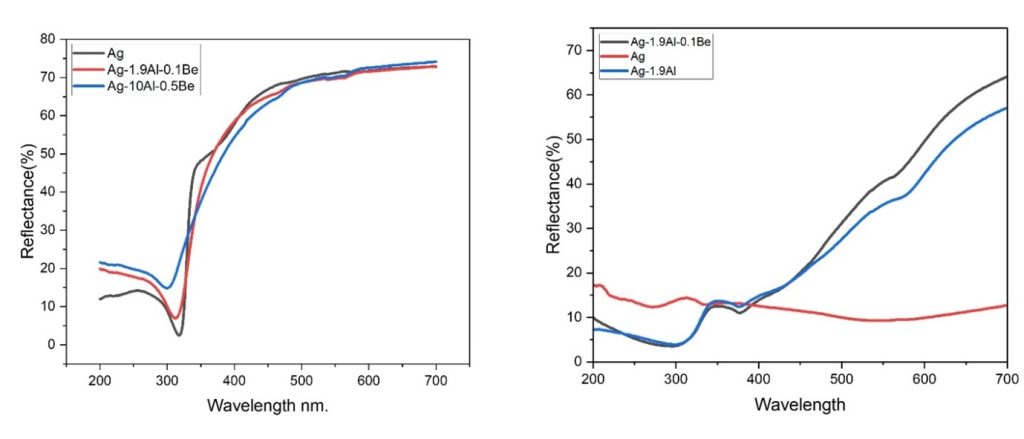
Figure.2 Reflectance of alloys (a) before tarnish test (b) after tarnish test

