Recent News
- Dr Jatis Kumar Dash to join the Editorial Board of Frontiers in Physics journal August 1, 2022
 Frontiers in Physics is a peer-reviewed scientific journal. It covers the entire field of Physics ranging from experimental to computational and theoretical physics. Assistant Professor Dr Jatis Kumar Dash from the Department of Physics has been invited to be a part of the Editorial Board of this prestigious journal. The multidisciplinary journal focuses on applied physics and has an impact factor of 3.560.
Frontiers in Physics is a peer-reviewed scientific journal. It covers the entire field of Physics ranging from experimental to computational and theoretical physics. Assistant Professor Dr Jatis Kumar Dash from the Department of Physics has been invited to be a part of the Editorial Board of this prestigious journal. The multidisciplinary journal focuses on applied physics and has an impact factor of 3.560.Dr Jatis Kumar Dash feels honoured to receive the invitation to join the Editorial Board. He finds it satisfying to get involved in scrutinising and reviewing manuscripts that concentrate on materials, experimental and condensed matter of Physics, and related devices. The process of reviewing these manuscripts enriches his knowledge in the subject domain. This helps him widen his research horison and explore novel ideas. The tenure of his role as an editor is not defined but is expected to last for five years.
Continue reading → - Published the first article under the Indo- Israel bilateral project July 29, 2022
 Research at the Department of Physics envisions future studies on the anisotropic properties across various planes of their reported MAPbBr3 crystals and identifying the better plane for efficient electrical contact in device applications. Assistant Professor Dr Sabyasachi Mukhopadhyay and his research scholar Kunchanapalli Ramya recently published the paper Room-Temperature Cost-effective In-situ grown MAPbBr3 Crystals and their Characterization towards Optoelectronic Devices in the journal Material Science and Engineering: B. The paper has an impact factor of 3.407. They have done this work in collaboration with Sr Satyajit, IIT- Bhilai. This is the first article published under the Indo- Israel bilateral project A Halide Perovskite-Based Photoanode for Oxygen Evolution Reaction Using a Molecular Diode in a Hybrid Nanometer Scale.
Research at the Department of Physics envisions future studies on the anisotropic properties across various planes of their reported MAPbBr3 crystals and identifying the better plane for efficient electrical contact in device applications. Assistant Professor Dr Sabyasachi Mukhopadhyay and his research scholar Kunchanapalli Ramya recently published the paper Room-Temperature Cost-effective In-situ grown MAPbBr3 Crystals and their Characterization towards Optoelectronic Devices in the journal Material Science and Engineering: B. The paper has an impact factor of 3.407. They have done this work in collaboration with Sr Satyajit, IIT- Bhilai. This is the first article published under the Indo- Israel bilateral project A Halide Perovskite-Based Photoanode for Oxygen Evolution Reaction Using a Molecular Diode in a Hybrid Nanometer Scale.Abstract
The paper reports the in-situ, room-temperature synthesis of methylammonium lead bromide CH3NH3PbBr3 crystals using N-methyl formamide as a source of methylammonium (MA+) ions during the crystallization process to explore the structural, dielectric, and electronic properties of CH3NH3PbBr3 crystals for optoelectronic applications. Optical absorption and radio-luminescence measurements affirm the direct bandgap nature of the crystals. Impedance spectroscopy measurements with various applied AC voltages within the 20 Hz – 10 MHz frequency range depict the influence of ionic motions on electrical transport across crystal planes. Researchers have extracted electrical transport parameters in CH3NH3PbBr3 crystals from the Nyquist plots, which we found to be distinctly varied wherein two different AC voltage amplitude regimes, broadly for 10 – 50 mV and 100 – 500 mV AC voltage range.
Explanation of the research
The wide approachability of our synthesis method lies in avoiding expensive precursor salts and eliminating the use of toxic solvents. We have obtained the MAPbBr3 crystals with improved thermal, optical, and dielectric properties that are used in optoelectronic devices, mainly in the applications of solar cells and photodetectors.
Continue reading → - DST-INSPIRE Subject Expert Committee Meeting July 18, 2022
 A two-day DST-INSPIRE Subject Expert Committee meeting was held on July 14 & 15 at SRM University-AP campus. Experts in the area of Physical Sciences from across the country gathered at the university to evaluate this year’s INSPIRE Fellowship applications in Physical Sciences.
A two-day DST-INSPIRE Subject Expert Committee meeting was held on July 14 & 15 at SRM University-AP campus. Experts in the area of Physical Sciences from across the country gathered at the university to evaluate this year’s INSPIRE Fellowship applications in Physical Sciences.INSPIRE Fellowship component offers 1000 Fellowships every year for carrying out doctoral degrees in both basic and applied sciences, including engineering and medicine, in the age group of 22-27 years.
The Chairperson of the Expert Committee in the area of Physical Sciences was Dr Dinakar Kanjilal, Professor, Inter-University Accelerator Centre (IUAC), New Delhi. “The fellowship ensures geographical distribution of excellence, and we look forward to more applicants from SRM AP”, Prof. Kanjilal said. Since its inception, SRM University-AP has INSPIRE Fellows as faculty members in the various departments of Sciences.
University Pro-Vice-Chancellor Prof D Narayana Rao, who also is the co-chair of the expert committee, said that it is gratifying to know that the PhD students have chosen to enrol in reputed universities and institutes. The applicants have chosen extremely accomplished scientists and faculty members as their research supervisors. “We are glad that 61 are girl students out of the 116 applications we received”, highlighted Prof D Narayana Rao. He expressed his happiness about the increasing number of women representation in Indian academia.
The other eminent scientists in the INSPIRE Fellowship selection committee included Member Secretary Dr Umesh K Sharma, Prof. Shikha Verma, Dr G. Vijaya Prakash, Dr AnandamayeeTej, Dr Rajendra Prasad Pant and Dr Arjit Chowdhuri.
Innovation in Science Pursuit for Inspired Research (INSPIRE)” is a flagship scheme of the Department of Science and Technology (DST), Government of India, which aims to attract meritorious youth to study basic and natural sciences at the college and university level and to pursue research careers in both basic and applied science areas including engineering, medicine, agriculture, and veterinary sciences.
Continue reading → - Highly stable ruthenium catalyst for hydrogen evolution reaction July 12, 2022
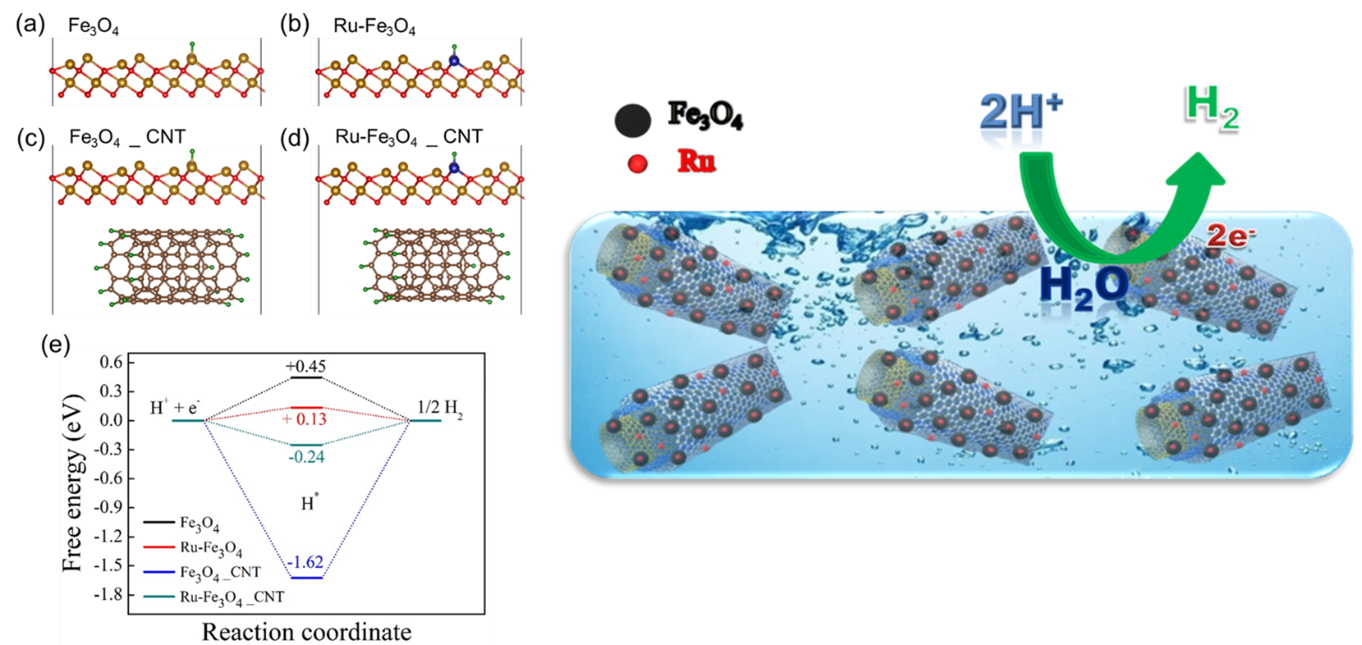
The Department of Physics is glad to announce that Prof Ranjit Thapa and his PhD scholar, Mr Samadhan Kapse, have published a patent titled “Highly Stable Ruthenium Single-Atom Catalysts on Fe3O4/MWCNTs for Hydrogen Evolution Reaction” (Application no. 202241006087). The research was done in collaboration with Ms Shwetha K R, Mr Shivanna M and Dr Nagaraju D H, from the Department of Chemistry, School of Applied Sciences, REVA University, Bangalore.
A Brief Description of the Research
In the current work, Fe3O4 nanoparticles were prepared by a simple chemical co-precipitation method under an inert atmosphere, and it was utilised for HER studies. Ru nanoparticles were profitably deposited over Fe3O4/MWCNTs modified glassy carbon electrode by the electrochemical deposition technique. The superior HER activity was achieved on Fe3O4/MWCNTs/Ru in 0.1M H2SO4 aqueous media. We demonstrated that synthesised electrocatalyst offers low over potential 101 mV to reach a current density of 10 mA cm-2 towards hydrogen evolution reaction. It displays exceptional stability and finds to be of no change in the HER activity despite 1000 cycles. It is emphasised that a small weight percentage of ruthenium in the prepared catalyst can replace high-cost platinum in renewable energy technologies.
Social Implications of the Research
Production of renewable energy has greater significance in the present situation owing to the impact of the depletion of non-renewable energy resources such as fossil fuels and the release of greenhouse gases into the atmosphere. Hydrogen has gained considerable interest as an energy storage and energy carrier due to its high energy density (146kJ/g), and its utilisation also eliminates pollution and toxicity. Several methods have been explored to produce molecular hydrogen. Among them, the electrolysis of water is the best way to produce high purity hydrogen from water. An excellent electrocatalyst is obligatory to liberate hydrogen gas effectively from water. It is known that Superior HER activity has been achieved using platinum (Pt) and Pt-based catalysts. Due to its high cost and low surplus, its expansion has been limited to the industrial scale. The research proposes that Ru-based catalysts can overcome these challenges.
DFT study is more effective to find the origin of catalytic activity in materials for designing highly promising catalysts for various catalytic reactions. The researchers expressed their gratitude to SRM University-AP for providing the required computational facility and support.
Continue reading → - 100% Erasmus Mundus Scholarship and MS opportunities in Europe July 5, 2022
 SRM University-AP has numerous success stories and student accomplishments to share with the world. What makes the story of Bennet Benny different is the magnitude of his winning and the miles he has crossed after setting foot to achieve his dreams. He has secured the much-coveted Erasmus Mundus Joint Masters Scholarship with a whopping sum of € 33,600 for two years. With the 100% EMJM scholarship, he can now pursue QuanTEEM Master’s across four different universities, each semester in one of these universities:
SRM University-AP has numerous success stories and student accomplishments to share with the world. What makes the story of Bennet Benny different is the magnitude of his winning and the miles he has crossed after setting foot to achieve his dreams. He has secured the much-coveted Erasmus Mundus Joint Masters Scholarship with a whopping sum of € 33,600 for two years. With the 100% EMJM scholarship, he can now pursue QuanTEEM Master’s across four different universities, each semester in one of these universities:University Bourgogne Franche-Comté (France)
Technische Universität Kaiserslautern (Germany)
Aarhus University (Denmark)
Moskovskiy Fiziko-Tekhnicheskiy Institut (Russia)Internship at JAIST, Japan -2019
Like every other student, Bennet joined the Bachelor’s degree programme at the Department of Physics in 2018 with an irrepressible desire to dive into the depths of Physics. His undying passion for grasping the subject’s nuances is an influential lesson for all students to emulate. When he was in the second year of his graduate studies, Bennet won the Sakura Science Internship under the supervision of Prof. Ryo Maezono at JAIST, Japan. The internship was funded by the Japan Science and Technology Agency (JST), a government funding agency. For him, this was an excellent opportunity to learn more deeply about quantum mechanics. It also helped him raise his awareness of computational physics, its advantages, uses and the latest research around it.
“I was able to interact with many international scholars and researchers at the Japan Advanced Institute of Science and Technology. It helped me learn about the various ways through which I could fund my higher education. Therefore, after returning to SRM University-AP, I could work in the necessary direction to build my profile accordingly.” Bennet remarked. The enormous lessons he learned there helped him publish a research paper under the guidance of Prof Ranjit Thapa and his PhD students in the Computational Physics laboratory. “The Department of Physics has always motivated me to reach greater heights”, he added.
NTU-India Connect Research Programme 2022
Bennet has also proved his mettle by securing yet another internship opportunity as part of the NTU-India Connect Research Programme 2022. He was selected to spend a semester (Spring term) at NTU, Singapore with a full fellowship during the final year of graduation. As a young researcher, he has gained immense exposure and experience in such a short period giving him a competitive edge to move further in the direction of his dreams. “It has always been my dream to pursue a research career in Physics”, he asserted.
Looking Forward to QuanTEEM Master’s
The QuanTEEM Master’s programme is based on Quantum technologies and provides an excellent opportunity to build a research network in multiple countries in the European Union. He aspires to gain a deeper understanding of quantum mechanics and wishes to use the knowledge to improve our civilisation. In the words of Bennet, “We should never let the fear of failure deter us from trying. I feel that to reach our best potential; we need to face the challenges in life rather than be disheartened by them. Hence, I would encourage my fellow students never to hesitate to seek new opportunities.”
Continue reading →

