Recent News
- Triumphant Trio: Dr Soumyajyoti and Scholars Make Strides in Physics April 22, 2024
 In a significant academic achievement, Dr Soumyajyoti Biswas, Assistant Professor and Scholars Ms Diksha and Mr Gunnemeda Eswar in the Department of Physics, has published an insightful paper in the prestigious Q1 journal, Physical Review E. The paper, titled “Prediction of depinning transitions in interface models using Gini and Kolkata indices,” presents a novel approach to understanding the complex phenomena of depinning transitions in physical systems.
In a significant academic achievement, Dr Soumyajyoti Biswas, Assistant Professor and Scholars Ms Diksha and Mr Gunnemeda Eswar in the Department of Physics, has published an insightful paper in the prestigious Q1 journal, Physical Review E. The paper, titled “Prediction of depinning transitions in interface models using Gini and Kolkata indices,” presents a novel approach to understanding the complex phenomena of depinning transitions in physical systems.The research introduces the use of Gini and Kolkata indices as predictive tools, offering a fresh perspective that could pave the way for new discoveries in the field. This publication not only underscores Dr. Biswas’s expertise but also enhances the university’s reputation as a hub for cutting-edge research.
The Department of Physics congratulates Dr. Biswas on this remarkable accomplishment and looks forward to the continued impact of his work on the scientific community.Abstract
The intermittent dynamics of driven interfaces through disordered media and its subsequent depinning for large enough driving force is a common feature for a myriad of diverse systems, starting from mode-I fracture, vortex lines in superconductors, and magnetic domain walls to invading fluid in a porous medium, to name a few. In this work, we outline a framework that can give a precursory signal of the imminent depinning transition by monitoring the variations in sizes or the inequality of the intermittent responses of a system that are seen prior to the depinning point. In particular, we use measures traditionally used to quantify economic inequality, i.e., the Gini index and the Kolkata index, for the case of the unequal responses of precritical systems.
The crossing point of these two indices serves as a precursor to imminent depinning. Given a scale-free size distribution of the responses, we calculate the expressions for these indices, evaluate their crossing points, and give a recipe for forecasting depinning transitions.We apply this method to the Edwards-Wilkinson, Kardar-Parisi-Zhang, and fiber bundle model interface with variable interaction strengths and quenched disorder. The results are applicable for any interface dynamics undergoing a depinning transition.
Explanation of Research in Layperson’s Terms
Many different physical systems, from cracking in materials to the movement of magnetic fields, share a common underlying behavior – they all involve an “interface” or boundary that moves through a disordered, or irregular, medium. As this interface moves, it often gets “pinned” or stuck in place by the disorder in the medium. However, as the driving force on the interface increases, there comes a point where the interface suddenly “depins” and starts moving much more freely. This transition from a pinned to a depinned state is called the “depinning transition” and it’s an important phenomenon in many areas of science and engineering.
In this work we have found a way to detect when this depinning transition is about to happen, before it actually occurs. We do this by looking at the sizes or magnitudes of the intermittent, or irregular, responses of the system as the driving force increases. Specifically, we use two measures that are traditionally used to quantify economic inequality – the Gini index and the Kolkata index. These give a way to track how “unequal” or unevenly distributed the sizes of the responses are. We found that when these two inequality measures cross each other, it serves as a precursor or early warning sign that the depinning transition is imminent. This is a powerful result because being able to predict when depinning will happen is very useful in fields like material science, superconductivity, and fluid dynamics, where controlling these phase transitions is important. By monitoring these inequality measures, one can may be able to forecast depinning events before they occur, which could lead to better design and control of these systems.
Title of Research paper in the Citation Format
Diksha, G. Eswar, and S. Biswas, Prediction of depinning transitions in interface models using Gini and Kolkata indices, Physical Review E 109, 044113 (2024).
DOI: https://doi.org/10.1103/PhysRevE.109.044113Practical Implementation or the Social Implications Associated with it
Prediction of imminent transition has implications in a wide range of disciplines, including stability of mechanical structures (buildings, aircraft, bridges, etc.), extraction of oil (fracking) to the largest scale of mechanical failure i.e., earthquakes.
Collaborations
This work is done with the PhD student Ms Diksha and the BSc student Mr Gunnemeda Eswar.
Future Research Plans
Future research plans include applications of the methods developed here to be applied to real-life physical structures for their stability analysis and predictions of impending catastrophes.
Pictures Related to the Research
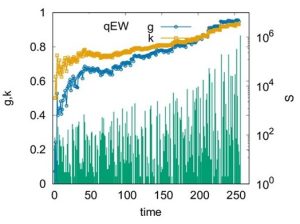
In this fig. the time series of avalanche sizes is shown along with the time variations of g and k for the quenched Edwards-Wilkinson model as a prototype. Here the crossing of g and k occurs prior to the depinning transition point (the last avalanche in the series). Therefore, the crossing of g and k can serve as a good indicator of an imminent depinning transition.
Continue reading → - Revolutionising Cooling Technology: Patent Granted for Innovative Fabric Design April 15, 2024
 The Department of Physics is thrilled to announce a remarkable milestone in sustainable technology. Dr Sabyasachi Mukhopadhyay, Associate Professor and Ms Sreelekha Bhuvaneswari, BSc student has been officially granted a patent for their groundbreaking invention: “A fibre material with moisture retention capacity with thermal tolerance and a method for manufacture of the same.”
The Department of Physics is thrilled to announce a remarkable milestone in sustainable technology. Dr Sabyasachi Mukhopadhyay, Associate Professor and Ms Sreelekha Bhuvaneswari, BSc student has been officially granted a patent for their groundbreaking invention: “A fibre material with moisture retention capacity with thermal tolerance and a method for manufacture of the same.”This pioneering material, distinguished by its Application Number 202141023375, stands at the forefront of cooling technology. It promises to enhance thermal regulation while prioritising environmental sustainability.
Dr Mukhopadhyay’s invention is poised to make significant contributions to the field of material science, offering a versatile solution that holds potential applications across various industries. The department extends its heartfelt congratulations to the duo on this significant achievement and looks forward to the positive impact his work will continue to have on our community and beyond.
Abstract:
The project, with the patent application number 202141023375, develops a methodology to design a fabric cloth that would replace the use of air conditioners. This cloth design is inspired by Saharan silver ants which regulate their body temperatures in the scorching desert heat and also from the cooling properties of clay. This research would significantly scale down the usage of AC and other cooling devices in warm places, thus reducing the use of electricity and emission of greenhouse gases to the environment. As this cloth would be environment friendly with long durability and cost-efficiency, Sreelekha hopes that this research would bridge the socioeconomic divide of haves and have-nots between communities.
Explanation of the Research in Layperson’s Terms :
Using room conditioning AC during these times, especially with lower temperatures is very risky. Virus spread with closed circulated air or prolonged cold temperatures while breathing, so thus using air conditioning is bad. With this in mind, we need an efficient way of cooling in the snoring temperatures. We propose developing a fabric that integrates clay (Its primary mineral is kaolinite; clay may be generally described as 40% aluminium oxide, 46% silicon oxide, and 14% water.) in its synthesis; clay is known for its cooling properties given enough water to retail and proper reflection techniques for heat throughout the fabric inspired from uniquely shaped hairs with triangular cross-sections as in arrangement employed by the Saharan silver ants. This model not only conserves energy compared to room cooling but also is very practical, wherein we can dampen the fabric with the least water and retain its cooling for long hours. This Fabric could be employed in a variety of settings, for example, in making carpets and curtains in a house to keep the room temperature regulated.
Practical Implementation and Social Implications:
The research would significantly scale down the usage of AC and other cooling devices in warm places, thus reducing the use of electricity and emission of greenhouse gases to the environment. As this cloth would be environment friendly with long durability and cost-efficiency, Sreelekha hopes that this research would bridge the socioeconomic divide of haves and have-nots between communities.
The Title of Patent in the Citation Format
“A fibre material with moisture retention capacity with thermal tolerance and a method for manufacture of the same” with Application Number: 202141023375
Collaborations:
Prof. Seeram Ramakrishna, Professor of Materials, National University of Singapore
Future Research Plans:
The forthcoming endeavour encompasses the commercialisation of this pioneering concept to render it widely accessible and applicable on a larger scale.
Continue reading → - Shining Bright: PhD Scholar Wins 2nd Prize at National Symposium March 20, 2024
 SRM University-AP is proud to congratulate Harsha, a brilliant PhD scholar from the Department of Physics, for her exceptional achievement at the National Symposium of Research Scholars on Metallurgy and Materials held at IIT Kanpur. Harsha secured the prestigious 2nd prize for her remarkable presentation titled “Tarnish Resistance Mechanism in Ag-4Cu-2.5Zn-1Ge Novel Alloy: A Comprehensive Experimental and Computational Analysis”.
SRM University-AP is proud to congratulate Harsha, a brilliant PhD scholar from the Department of Physics, for her exceptional achievement at the National Symposium of Research Scholars on Metallurgy and Materials held at IIT Kanpur. Harsha secured the prestigious 2nd prize for her remarkable presentation titled “Tarnish Resistance Mechanism in Ag-4Cu-2.5Zn-1Ge Novel Alloy: A Comprehensive Experimental and Computational Analysis”.The symposium, organised at IIT Kanpur, brought together some of the brightest minds in the field of metallurgy and materials research. Harsha’s presentation stood out among the impressive array of research papers, showcasing her expertise and dedication to pushing the boundaries of scientific exploration.
Her presentation focused on the intricate mechanism of tarnish resistance in the Ag-4Cu-2.5Zn-1Ge novel alloy. Through a comprehensive analysis that combined experimental and computational approaches, Harsha shed light on the factors influencing tarnish resistance in this unique alloy composition. Her findings have significant implications for the development of advanced materials with enhanced durability and longevity.
The symposium, which congregates the brightest minds in the field, was the perfect platform for Harsha to showcase her extensive research. Her work, which provides significant insights into the tarnish resistance mechanisms of novel silver alloys, has been recognised for its depth and scientific rigour.
The recognition received by Harsha at this prestigious national symposium is a testament to her exceptional research capabilities and the quality of education and mentorship provided at SRM University-AP. It highlights the university’s commitment to nurturing talent and fostering an environment conducive to groundbreaking research.
SRM University-AP extends its warmest congratulations to Harsha for this remarkable achievement. Her success serves as an inspiration to fellow students and researchers, encouraging them to pursue excellence in their respective fields of study.
Continue reading → - SOFEEM-2024: A National Conference on Functional Energy and Electronic Materials March 7, 2024

SRM University-AP hosted a national conference on “Status and Opportunities in Functional Energy and Electronic Materials” (SOFEEM-2024) on March 02-03, 2024. The conference was organised by the Department of Physics and was supported by the Department of Science and Technology, Government of India. The conference brought together about 100 participants from various reputed institutions, national laboratories, and universities from India. They shared their research outcomes and discussed the various frontier energy technologies and smart electronic devices.
The main theme of the conference was to boost the development and optimal use of renewable energy and to promote sustainable energy and intelligent electronics. The conference focused on fostering collaboration among researchers and practitioners to explore cutting-edge developments in these domains and pave the way for innovative solutions. A diverse array of topics was covered, including:
- Synthesis and processing of novel energy and electronic materials
- Modelling of low-dimensional materials
- Materials and catalysts for green hydrogen production
- Electrolyte and electrode materials for high-performance batteries and fuel cells
- Functional materials for supercapacitors
- Device fabrication using novel energy materials for energy conversion and storage
- Low-dimensional materials in electronics and photonics
The conference was inaugurated by Prof. Manoj K Arora, Vice Chancellor, who welcomed the guests and the delegates and highlighted the university’s achievements and vision. He also emphasised the importance of interdisciplinary research and innovation in addressing the global energy and environment challenges.

The conference featured keynote lectures, invited talks, oral presentations, and poster sessions by experts and young researchers. Distinguished Keynote speakers from IIT Madras, along with Invited speakers from prestigious institutions such as the Indian Institute of Science (IISC) Bangalore, IIT Tirupati, IIT Hyderabad, University of Hyderabad, IISER Bhopal, and the CSIR – Central Electrochemical Research Institute (CECRI) in Tamil Nadu, among others from esteemed state universities, shared their groundbreaking research. The conference provided a good platform for the participants to interact with the eminent researchers to enrich and share their knowledge in their respective fields of research and engage in potential collaboration to develop innovative technologies in the areas of sustainable energy, advances in materials research and computational sciences. In addition, research scholars from nearby universities (such as VIT-AP, Vignan University, Acharya Nagarjuna University, and SRM-IST) participated in the conference and showcased their current research works.
The conference concluded with awards for the best poster and oral presentation by research scholars, celebrating their significant contributions to the field. The conference concluded with a valedictory session, where the best oral and poster presentations were awarded, and feedback from the participants was collected. The conference was well-received by the attendees, who appreciated the quality and diversity of the scientific discussions, as well as the university’s hospitality and facilities.
Continue reading → - 3rd ICMG Inauguration: Emerging Interfaces in Materials Genome February 29, 2024
 The third chapter of the biennial International Conference on Materials Genome (ICMG) was inaugurated at SRM University-AP, India on February 22, 2024, in the august presence of Prof G P Das, Research Institute for Sustainable Energy, India, Prof Yoshiyuki Kawazoe, Tohoku University, Japan, Prof Puru Jena, Virginia Commonwealth University, USA, Prof Jer Lai Kuo, Dr R Premkumar, Registrar, SRM University AP, Prof Ranjit Thapa, Dean-Research, Dr Pranab Mandal, Associate Professor & Head of the Department, Physics, Dr Mahesh Ravva, Associate Professor, Chemistry, Dr Pradyut Kumar Sanki, Associate Professor, Electronics and Communication Engineering and 150+ dignitaries across the world.
The third chapter of the biennial International Conference on Materials Genome (ICMG) was inaugurated at SRM University-AP, India on February 22, 2024, in the august presence of Prof G P Das, Research Institute for Sustainable Energy, India, Prof Yoshiyuki Kawazoe, Tohoku University, Japan, Prof Puru Jena, Virginia Commonwealth University, USA, Prof Jer Lai Kuo, Dr R Premkumar, Registrar, SRM University AP, Prof Ranjit Thapa, Dean-Research, Dr Pranab Mandal, Associate Professor & Head of the Department, Physics, Dr Mahesh Ravva, Associate Professor, Chemistry, Dr Pradyut Kumar Sanki, Associate Professor, Electronics and Communication Engineering and 150+ dignitaries across the world.ICMG is set up to nurture and promote research and development activities in computational materials in association with Asian Consortium on Computational Materials Science (ACCMS), Jawaharlal Nehru Centre for Advanced Scientific Research, Bangalore (JNCASR), National Chemical Laboratory, Pune (NCL), Virginia Commonwealth University, USA, Indian Institute of Technology, Madras (IIT-M), Indian Institute of Technology, Bombay (IIT-B), TCG-Crest, Kolkata.
In his inaugural speech, Dr R Premkumar delved into the vision of SRM University-AP as a new-age research-centric university. Within 7 years of inception, SRM University-AP proudly harbours 20 departments across 3 schools, providing education to 8500 students. The university aspires to bring quality education to 15000 students in the near future. The research environment of the university is no less than excellent. The university strives to contribute to society through outstanding research activities. The premier faculty pool of the university with 100% PhD and international academic and industry exposure and the zeal to do quality research makes it a chosen haven for achievers. SRM University-AP supports its faculty and students in research by providing Seed Funding, Research Grants, Financial Assistance for Publications and Patents, Conference Aids and state-of-the-art research facilities. “The university’s mission is to create a newer generation who could substantially contribute to the academia and industry. Hence, we encourage our students with equal importance to take entrepreneurship, higher studies and/or Placement support as their future endeavours”, said Dr R Premkumar.
In his keynote speech, Prof. G P Das congratulated the University for the successful arrangement of the 3rd International Conference on Materials Genome. In the year 2000, the Asian consortium was born from a tea table discussion and has come a long way since. This consortium has become 25 years old, and they have been utilizing human as well as computational resources in different countries in the Asian region. The year 2024 marks the 60th year of the two most important milestone developments in the field of materials simulation – (i) Density Functional Theory by Walter Kohn and (ii) the landmark paper by Aneesur Rahman who is considered the founding father of computer simulation and molecular dynamics. It is exciting to note how the research in this domain has evolved in the years between. “One of the most recent additions to this is the marriage between materials science and computer science. This is also termed materials informatics or materials genomics. Big data, deep neural networks, reinforcement learning etc. are becoming more and more familiar tools for materials physicists and chemists. An increasingly large number of papers are appearing in the most prestigious peer-reviewed journals on the data-centric approach to discover as well as predict novel energy materials, catalytic materials, magnetic & spintronic materials, various kinds of functional materials and more recently topological materials,” asserted Prof Das.
Prof. Das further emphasised that there are a large number of areas that are being investigated with the aid of modern technologies like AI-ML. For example, the space group of a complex geological sample can be predicted just by looking at its X-ray or Neutron diffraction data using deep neural networks which was conventionally used to be done by Rietveld analysis, but now such inverse problems can be dealt with AI-ML. Such data-centric pattern-recognition approaches and their triumphs in predicting the behaviour of hitherto unknown areas e.g. cryptography, agriculture, and even criminology open up a bright new future ahead of us. Prof. Das concluded his speech by reminding us that we are now far ahead of our conventional ways of studying. The emerging ‘interface areas’ between disciplines such as biology and physics, economics & physics, metallurgy & physics, electronics & physics, mathematics & statistics, and many other suggests an enormous leap in computational approaches with advances in experimental measurements.
The Sub-themes of the conference for this year are Quantum Mechanics/Machine Learning Approach, High-throughput Computation and Machine Learning aided discovery of Materials, on which 6 keynote speeches and 46 invited lectures will be delivered in the coming 3 days. It is indeed heartening to see the conglomeration of so many stalwarts covering broad areas in Physics, Chemistry and Materials Science.
Continue reading →












